During the health care pandemic and beginning in March 2020, the Centers for Medicare & Medicaid Services (CMS) suspended the regular survey process and announced its plan to conduct limited surveys that focused on infection prevention and control issues.[i] CMS has publicly reported the results of these limited services each month through a link on its website Nursing Home Compare (now Care Compare[ii]). The Center for Medicare Advocacy has analyzed these survey reports monthly and reported our findings in a series of CMA Alerts and Reports.
In August, CMS encouraged states to resume conducting standard (annual) and complaint surveys, to the extent possible,[iii] and is now reporting these survey findings through the same link on its public website. The CMS report describes the cumulative infection control deficiencies (F-880) that have been cited following infection control surveys, annual surveys, and complaint surveys since March. While an increasing number of infection control deficiencies are cited, most deficiencies continue to be classified as not causing residents any harm.
November 2020 Data
Although CMS has generally posted data on the last Wednesday of each month, it posted its seventh cumulative data release on November 18, reflecting deficiencies cited from March through October, as reported by state survey agencies to CMS. The cumulative data release for November includes, for the first time, deficiencies that were cited in much earlier months. For example, the July data release (covering deficiencies reported through June) indicated that 347 infection control deficiencies had been cited; the November data release (as reported below and covering deficiencies reported through October) indicates that 972 infection deficiencies were cited, as of June.
According to the November release, 4052 facilities have been cited with at least one infection control deficiency since March. A considerably higher percentage of surveys (nearly 10 percent) cited infection control deficiencies, but a lower percentage (6.38 percent) of the deficiencies were cited as immediate jeopardy, compared to the October cumulative release.
| Date of CMS release | Total number of surveys reported | Total number (percent) of infection control deficiencies cited | Total number (percent) of immediate jeopardy deficiencies cited |
| June 4 | 5,724 | 163 (2.8%) | 1 (1.0%) |
| June 24 | 9,899 | 262 (2.6%) | 4 (1.5%) |
| July 29 | 16,987 | 347 (2.0%) | 22 (6.6%) |
| August 26 | 25,593 | 556 (2.2%) | 48 (8.6%) |
| September 30 | 32,681 | 777 (2.4%) | 75 (9.7%) |
| October 28 | 40,144 | 1,104 (2.8%) | 112 (10.1%) |
| November 18 | 52,680* | 4,918 (9.34%) | 314 (6.38%) |
*A total of 61,820 surveys were conducted but more than 9,000 were duplicates (for example, some surveys were coded as both complaint and infection control) so that only 52,680 distinct surveys were actually conducted. The 61,820 duplicated surveys included 37,772 focused infection control surveys, 873 standard surveys, and 23,175 complaint surveys.
Infection Control Deficiencies, National Data
These 52,680 unduplicated surveys cited a total of 4918 infection control deficiencies at 4052 nursing facilities.
Focused infection control surveys cited the largest number of infection control deficiencies.
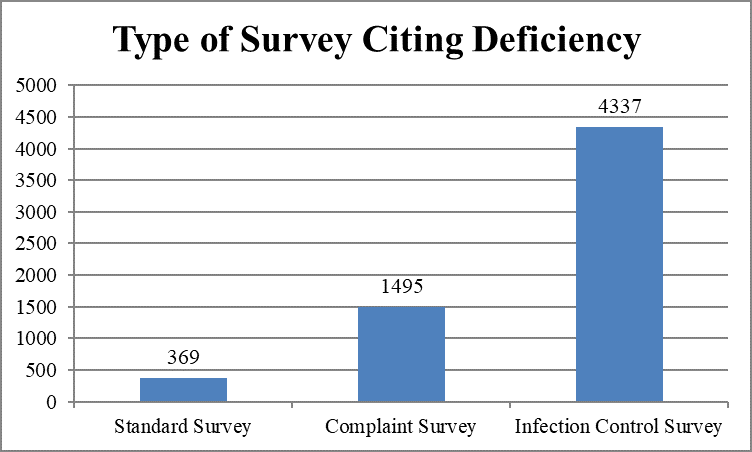
As shown below, most infection control deficiencies (4580, 93 percent) were classified as “no actual harm, with the potential for minimal harm” (“no harm”) – categories D, E, and F.
Scope and Severity of Deficiencies Cited, Cumulative
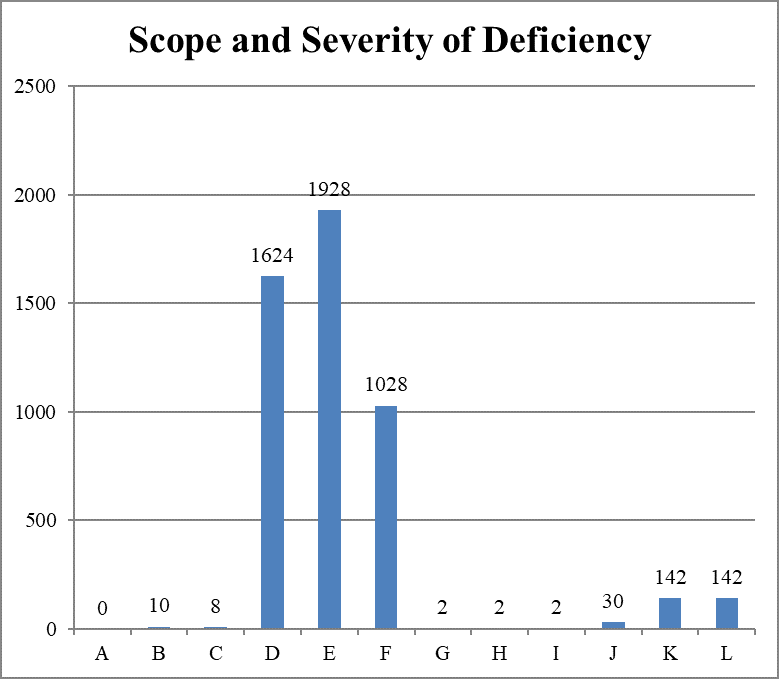
The seventh cumulative report documents a large increase in infection control deficiencies. However, most of them (4580, 93 percent) were classified as no harm; only 314 of these deficiencies (6.38 percent) were classified as immediate jeopardy.
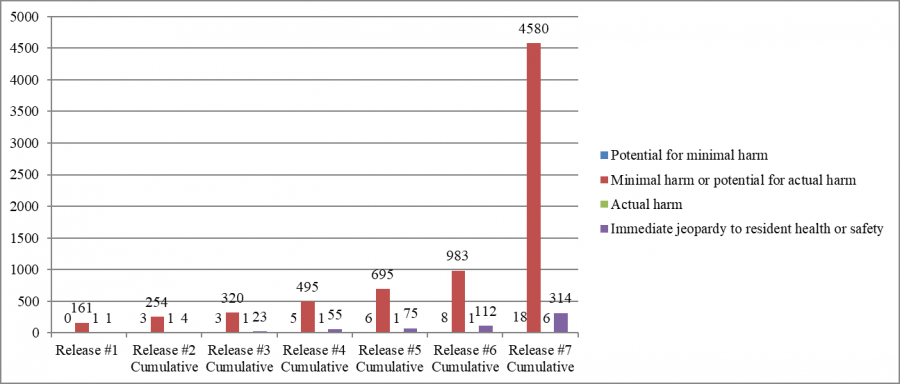
Deficiencies Cited, by Monthly Cumulative Report
Scope and Severity, Cumulatively, by Release Number
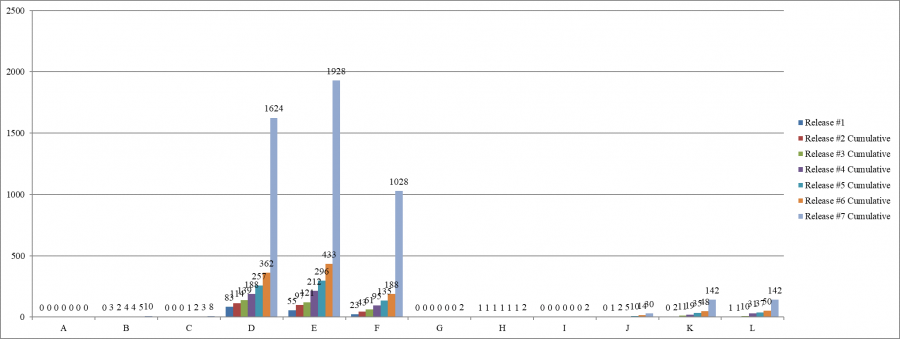
The largest number of infection control deficiencies was cited in June, 972 deficiencies; the smallest number, in March, 322 deficiencies.
Infection Control Deficiencies, by Month
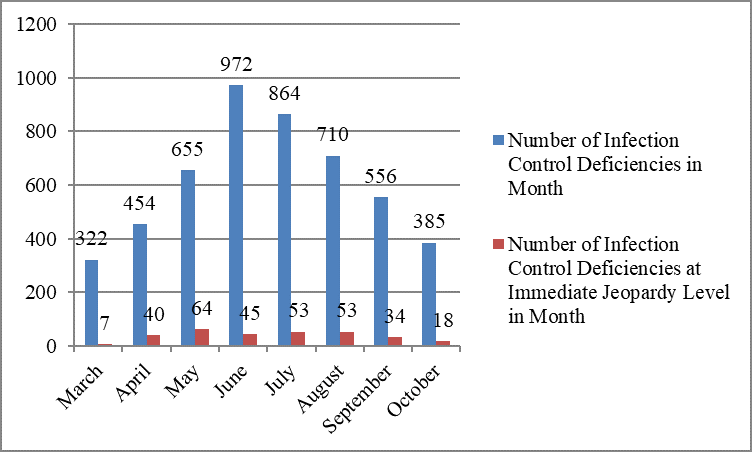
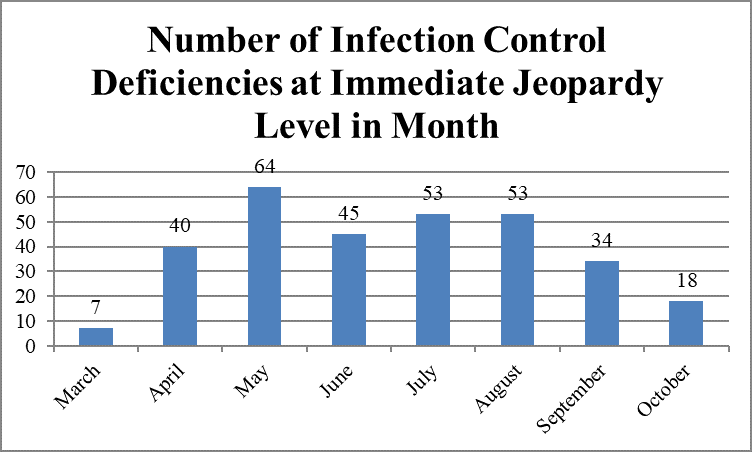
However, the largest number of immediate jeopardy deficiencies was cited in May, 64; the smallest number, in March, seven.
Immediate Jeopardy Infection Control Deficiencies, by Month
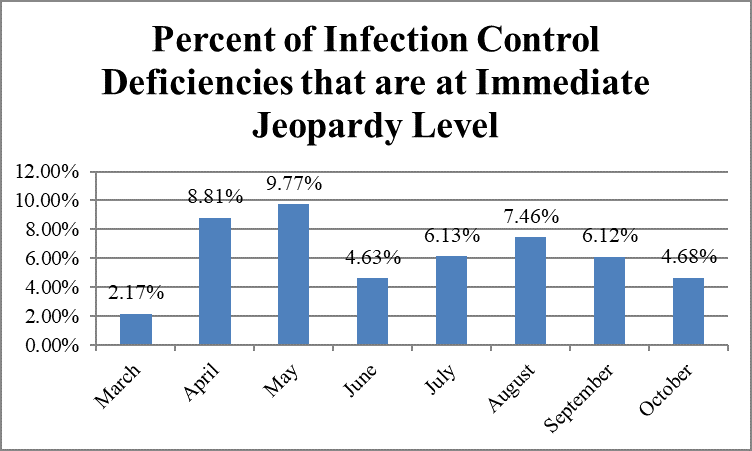
The largest percentage of deficiencies classified as immediate jeopardy was cited in May, 9.77 percent; the smallest percentage, in March, 2.17 percent.
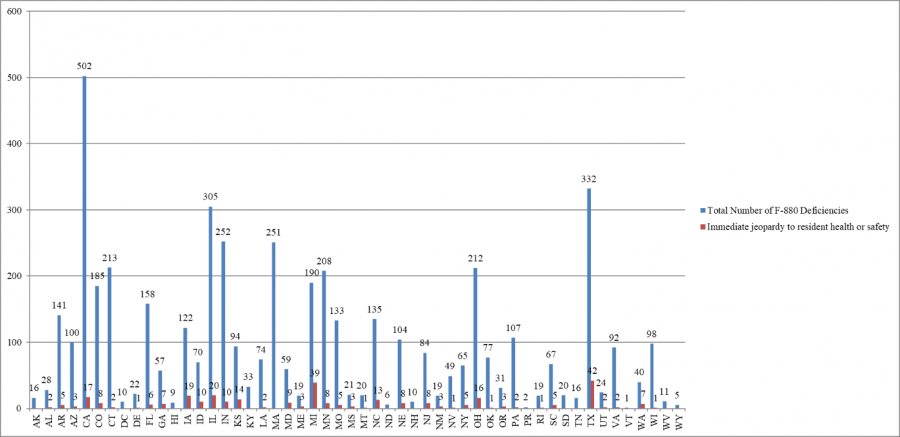
Infection Control Deficiencies, State Data
The November data release, for the first time, reports that every state cited at least one infection control deficiency since March.
The seventh cumulative data release indicates that California has cited the largest number of infection control deficiencies – 502. Until this data release, Texas had been reported to have cited the largest number of infection control deficiencies.
Cumulative Infection Control Deficiencies by State
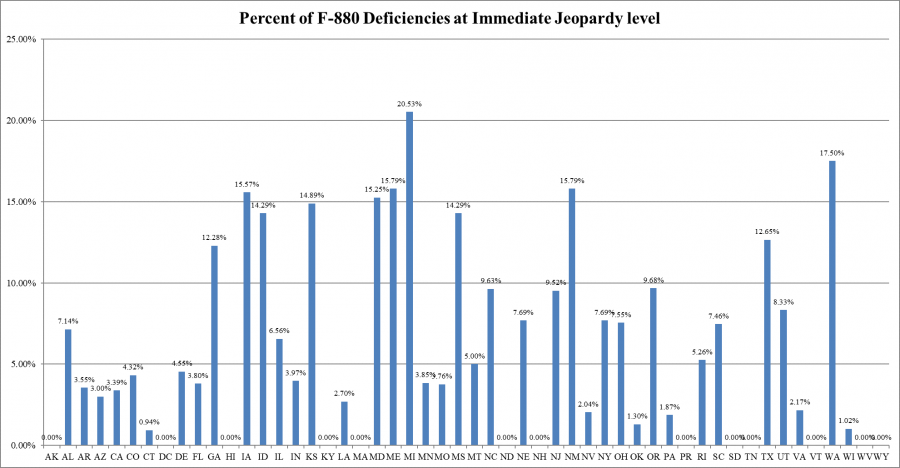
However, the percentage of infection control deficiencies cited as immediate jeopardy by each state ranged from zero percent in 11 states (Alaska, Hawaii, Kentucky, Massachusetts, New Hampshire, North Dakota, South Dakota, Tennessee, Vermont, West Virginia, and Wyoming), Puerto Rico, and the District of Columbia to 20.53 percent (Michigan).
Percent of Infection Control Cited as Immediate Jeopardy, by State
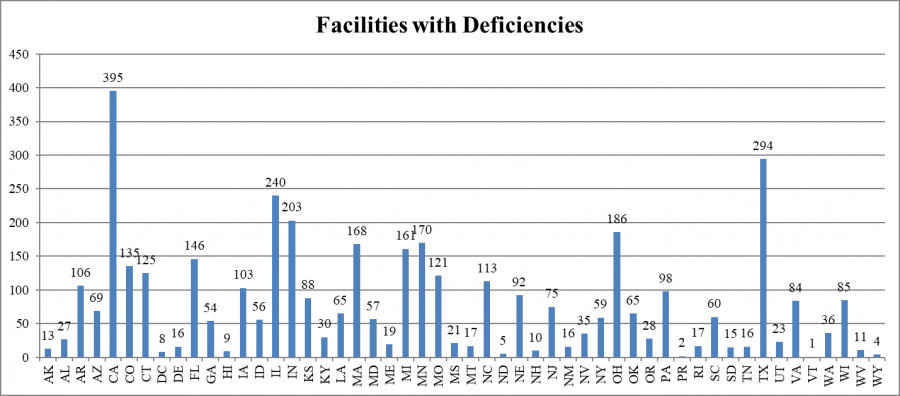
States cited various numbers of facilities with infection control deficiencies. California cited the largest number of facilities (395), while Vermont cited one. (California has more than 1200 nursing facilities; Vermont, 38 nursing facilities.)
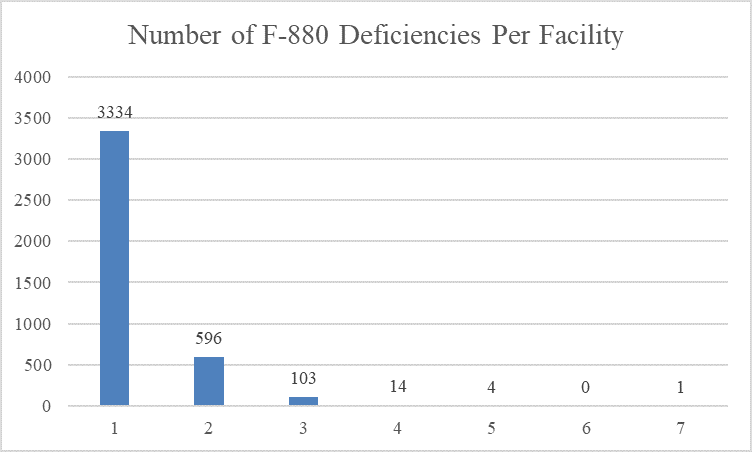
Four thousand fifty-two (4052) facilities were cited with an infection control deficiency. Most facilities (3334 facilities) were cited with a single infection control deficiency; 596 facilities were cited with two infection control deficiencies; 103 facilities were cited with three infection control deficiencies; 14 facilities were cited with four infection control deficiencies; four facilities were cited with five infection control deficiencies; and one facility was cited with seven infection control deficiencies.
Conclusion
For the first time, all states cited at least one infection control deficiency. Eleven states have not cited any immediate jeopardy deficiencies in infection control. Nonetheless, as throughout the pandemic, most deficiencies have been classified as “no harm.”
Dec. 17, 2020 – M. Edelman, T. Edelman
[i] CMS, “Suspension of Survey Activities,” QSO-20-12-All (Mar. 4, 2020), https://www.cms.gov/files/document/qso-20-12-all.pdf; CMS, ‘Prioritization of Survey Activities,” QSO-20-20-All (Mar. 23, 2020), https://www.cms.gov/files/document/qso-20-20-all.pdf
[ii] Center for Medicare Advocacy, “CMS Replaces all Eight Care-Specific Websites with Single Website: Care Compare” (CMA Alert, Dec. 10, 2020), https://medicareadvocacy.org/cms-replaces-all-eight-care-specific-websites-with-single-website-care-compare/
[iii] CMS, “Enforcement Cases Held during the Prioritization Period and Revised Survey Prioritization,” QSO-20-35-ALL (Aug. 17, 2020), https://www.cms.gov/files/document/qso-20-35-all.pdf
